Research
Chemical Bonding & Conceptual Method Development
Understanding the connection between electronic structure and material properties is a principal challenge in chemistry and material science. Useful insights into chemical bonding allow for chemical intuition, which guides and can dramatically accelerate experimental efforts.
We are interested in the adaptation, evaluation and development of chemical concepts to new challenging conditions. For example, we study universally applicable chemical descriptors (such as atomic radii and electronegativity), and methods capable of analyzing chemical bonding in molecules, in extended forms of matter (polymers and crystals), and in high pressure conditions. Much of our work in this field connects to the development of “Experimental Quantum Chemistry” (EQC), a framework through which the energy of any transformation (such as a bond formation) is described as a sum of terms that can be interchangeably obtained by computation or experiment.
In addition to facilitating for chemical rationales across different fields of disciplines and thermodynamic conditions (high pressure, low temperature, long time-scales), our research is generating promising input data for future machine learning approaches aimed at high throughput material discovery.

Research Highlights:
- A Density Functional Theory for the Average Electron Energy
S. Racioppi, P. Lolur, P. Hyldgaard, M. Rahm
J. Chem. Theory Comput. 19, 799–807, 2023 - M. Rahm, T. Zeng, R. Hoffmann, Electronegativity Seen as the Ground State Average Valence Electron Binding Energy, J. Am. Chem. Soc, 141,342-351, 2019 [A new scale of electronegativity]
- M. Rahm, R. Cammi, N. W. Ashcroft, R. Hoffmann, Squeezing all Elements in the Periodic Table: Electron Configuration and Electronegativity of the Atoms under Compression, J. Am. Chem. Soc.,141, 10253-10271, 2019 [The Periodic Table at 0-300 GPa]
- M. Rahm, R. Hoffmann, Distinguishing Bonds, J. Am. Chem. Soc., 138, 3731-3744, 2016 [Introduction to the Q-descriptor, shown in figure]
- M. Rahm, R. Hoffmann, N.W. Ashcroft, Atomic and Ionic Radii of Elements 1-96, Chem. Eur. J. 22, 14625-14632, 2016
Computational Astrobiology
What is the molecular basis of life, and how did life originate? These are fundamental questions that ultimately will involve systems of extreme complexity. To help evaluate which initial chemistry might have given rise to such complexity, we are studying one of the Universe’s simplest and most ubiquitous molecules.
Hydrogen cyanide, HCN, is believed to be a key ingredient for prebiotic chemistry, and a versatile building block for the construction of biomolecules. However, the chemical structure of HCN-based polymers remains an open question. There are significant challenges associated with the characterization of polymeric HCN, and highly complex mixtures of soluble and insoluble materials are typically generated in laboratories.
To tackle this challenge, we are using a combination quantum chemistry and theoretical condensed matter physics methodology to explore well-defined HCN-based materials and polymerization mechanisms. It is possible that such structures might form under kinetic controlled conditions (low temperature) and over long time-scales. To evaluate how HCN chemistry plays into observations we are collaborating with planetary scientists, whose particular focus is Saturn’s moon Titan. Titan can, in some respects, be considered a frozen over version of the early Earth, and the former provides a natural laboratory for studying prebiotic chemistry occurring under cryogenic conditions. One of the most abundant products of Titan’s atmospheric chemistry is HCN, where it contributes to the formation of not yet well-understood photochemical hazes.
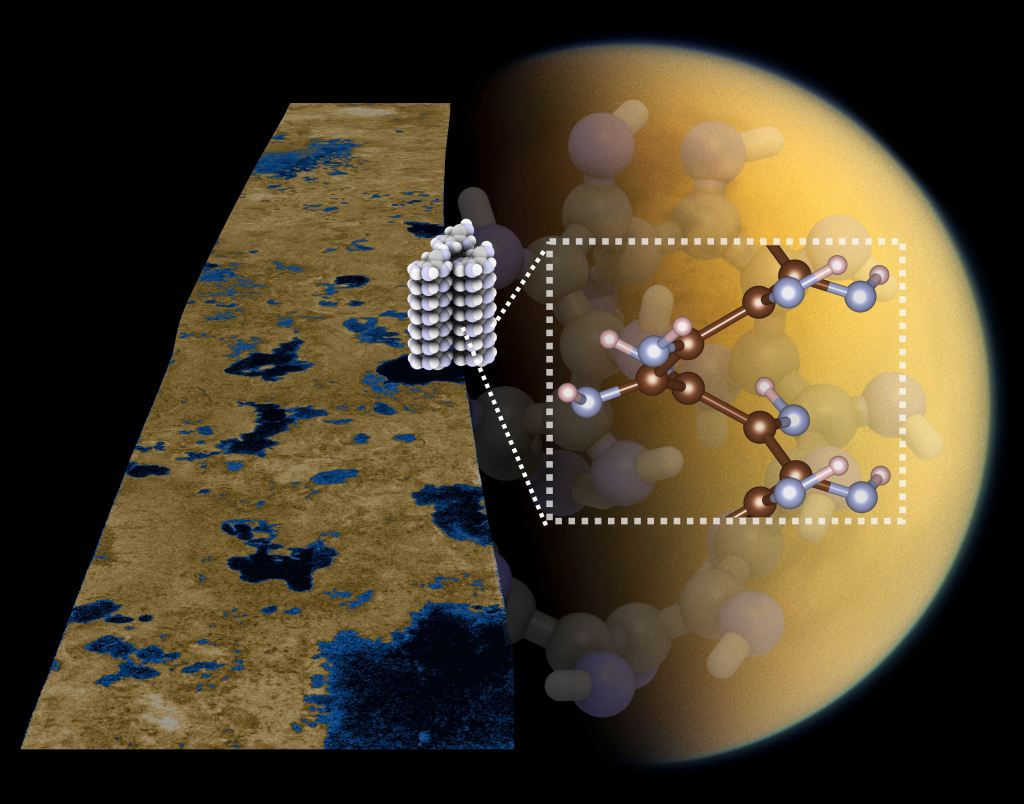
Research Highlights:
- Crossroads at the Origin of Prebiotic Chemical Complexity: Hydrogen Cyanide Product Diversification
H. Sandström, M. Rahm
J. Phys. Chem. A.,127, 20, 4503–4510, 2023 - H. Sandström, M. Rahm, Can Polarity-inverted Membranes Self-Assemble on Titan?, Sci. Adv. 6, eaax0272, 2020
- M. Rahm, J. I. Lunine, D. A. Usher, D. Shalloway, Polymorphism and Electronic Structure of Polyimine and its Potential Significance for Prebiotic Chemistry on Titan, PNAS, 113, 8121-8126, 2016
Quantum Computation of Chemistry
We develop quantum algorithms for performing quantum chemical calculations on quantum computers. To do so, the group work in close collaboration with experts in quantum technology within the Wallenberg Centre for Quantum Technology (WACQT) at the Department of Microtechnology and Nanoscience at Chalmers. WACQT is a 12-year initiative (2018-2029) aimed to bring Swedish academia and industry to the forefront of Quantum Technology. At Chalmers, the focus is to build a superconducting quantum computer and simulator and to explore useful applications of quantum computing.

Research Highlights:
- The Electron Density: A Fidelity Witness for Quantum Computation
M. Skogh, W. Dobrautz, P. Lolur, C. Warren, J. Biznárová, A. Osman, G. Tancredi, J. Bylander, M. Rahm
Chem. Sci., 15, 2257-2265, 2024 - Reference-State Error Mitigation: A Strategy for High Accuracy Quantum Computation of Chemistry
P. Lolur, M. Skogh, W. Dobrautz, C. Warren, J. Biznárová, A. Osman, G. Tancredi, G. Wendin, J. Bylander, M. Rahm
J. Chem. Theory Comput. 19, 783–789, 2023